This was a 20 year old primi gravida on treatment for long standing seizure disorder ; she was on tab carbamazepine 200 mgm twice daily for a long time . The scan was done around 26 weeks of gestation.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2970517/ – this link discusses teratogenic effects of anti epileptic drugs.
Carbamazepine (CBZ) is an iminostilbene derivative used primarily in the management of epilepsy and trigeminal neuralgia.
Observational studies have shown that the use of CBZ in pregnancy is associated with a higher risk of MCMs ( Major Congenital Malformations ) in the exposed offspring.
Other investigations using different methodologies have failed to find an increased statistical risk of MCMs due to CBZ exposure .
Morrow et al. reported 20 MCMs among 900 CBZ-exposed pregnancies, which is a prevalence rate of 2.2% with 95% CI: 1.4–3.4 . This is the lowest risk for MCMs amongst all AED monotherapy exposures reported in the literature . When dosage was compared between healthy and malformed infants, there was no statistical significance (p = 0.56), suggesting that genetic factors are interacting with the CBZ exposure contributing disproportionately to the risk for MCMs.
Association between CBZ exposure and specific MCMs :
increased risk of orofacial clefts
cardiac malformations
increased risk for NTDs
The images are given below.
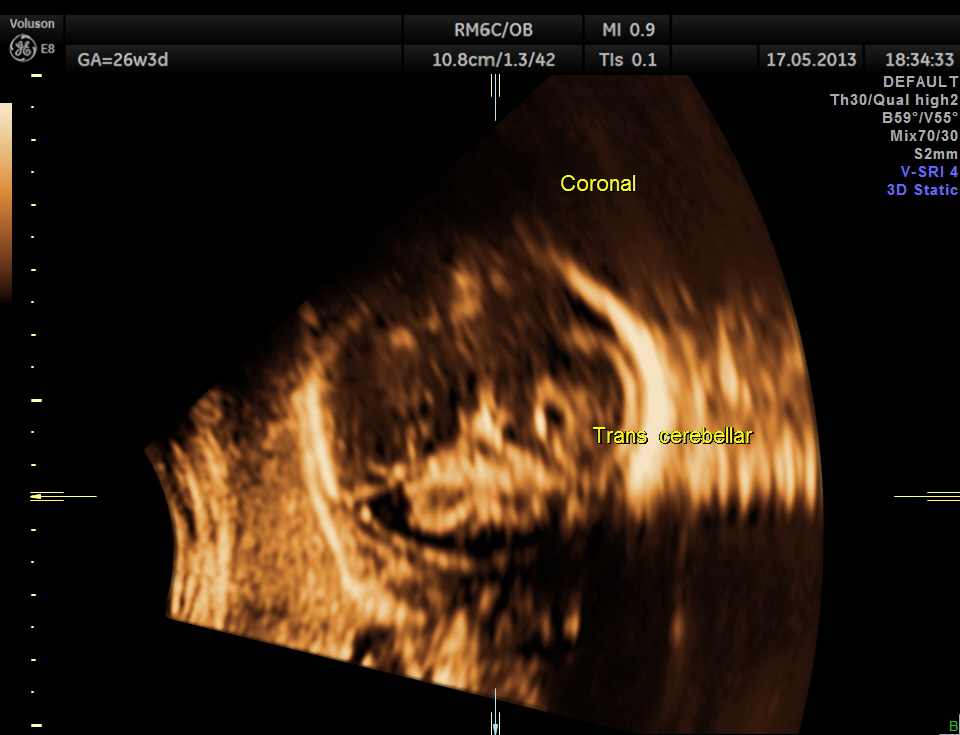
Cerebellum appears relatively smaller and corresponds to a GA of 22 to 23 weeks. Usually the cerebellum corresponds very closely to the GA and would be very useful to follow in cases of IUGR.
The following are 3 D reconstructed sagittal and coronal images .
The following images show the face and the palate.
The following images show the sections of the heart.
The following images show the spine.
The abdominal images are given below.
The limbs are shown below.
This fetus did not show any oro-facial clefts ; heart and spine appeared normal.
Cerebellum was < 2.3 %tile , suggestive of possible cerebellar hypoplasia. Unfortunately this patient was lost for follow up .
The following link gives more information about use of carbamazwpine in pregnancy.
http://www.drugs.com/pro/carbamazepine.html#_E434F86B-5F32-6E00-818D-B05080BAA2A6
Usage in Pregnancy
Carbamazepine can cause fetal harm when administered to a pregnant woman.
Epidemiological data suggest that there may be an association between the use of Carbamazepine during pregnancy and congenital malformations, including spina bifida. There have also been reports that associate Carbamazepine with developmental disorders and congenital anomalies (e.g., craniofacial defects, cardiovascular malformations, hypospadias and anomalies involving various body systems). Developmental delays based on neurobehavioral assessments have been reported. In treating or counseling women of childbearing potential, the prescribing physician will wish to weigh the benefits of therapy against the risks. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
Retrospective case reviews suggest that, compared with monotherapy, there may be a higher prevalence of teratogenic effects associated with the use of anticonvulsants in combination therapy. Therefore, if therapy is to be continued, monotherapy may be preferable for pregnant women.


























Thanks for Sharing the case. All slides are beautiful .
LikeLike
Thanks sir
LikeLike
many thanks nice case
LikeLike
T
LikeLike
Very nice sir… thank you…
LikeLike
Thanks
LikeLike
quite helpful
LikeLike